Your 30 Years of FDA Compliance Are Digitally Invisible — And AI Is Why It Matters Now
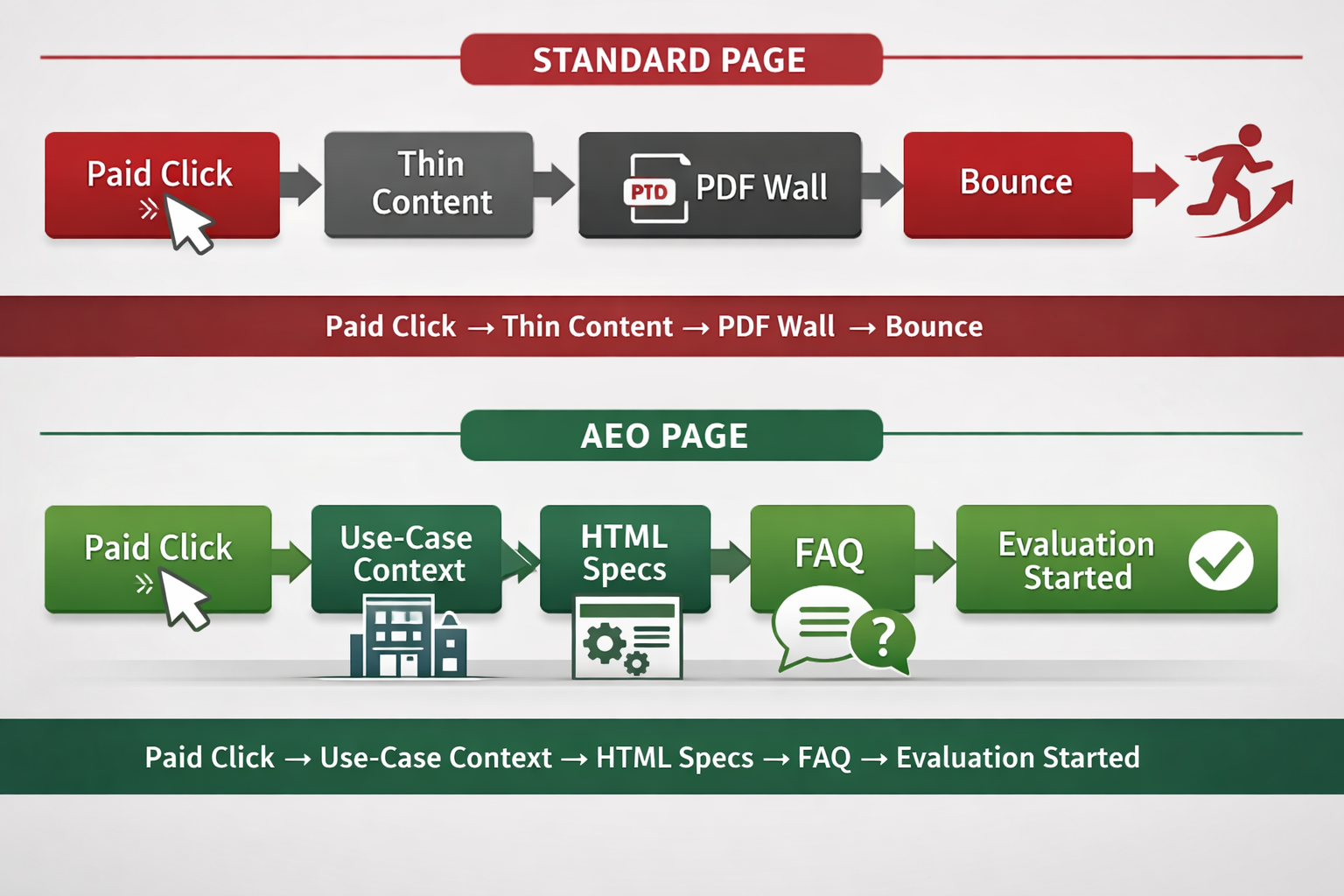
Medical Device Manufacturing · AEO Strategy · Regulatory Visibility Legacy medical device manufacturers have built decades of regulatory credibility. In 2026, that credibility is largely undetectable by the AI systems that clinicians, procurement teams, and developers now use to evaluate suppliers. Executive Brief As of February 2, 2026, the FDA officially completed its transition from … Read more